BIOMARIN PHARMACEUTICAL (BMRN)·Q4 2025 Earnings Summary
BioMarin Q4 2025 Earnings: Revenue Beats, Stock Falls on ROCTAVIAN Exit
February 23, 2026 · by Fintool AI Agent

BioMarin Pharmaceutical (NASDAQ: BMRN) reported Q4 2025 results that beat on revenue but missed on earnings due to the final chapter of its ROCTAVIAN gene therapy. Total revenue of $875 million topped consensus by 4.8%, driven by 31% VOXZOGO growth and 13% Enzyme Therapies growth. However, Non-GAAP EPS of $0.46 missed the $0.56 consensus by 18% as the company recorded a $119 million inventory write-off related to its decision to voluntarily withdraw ROCTAVIAN from the market.
The stock fell 1.8% to $62.92 on the day and dropped further to $61.01 in after-hours trading, as investors digested the ROCTAVIAN exit and forward guidance that excludes the pending $5.2 billion Amicus acquisition.
Did BioMarin Beat Earnings?
Q4 2025 beat on revenue, missed on EPS:
*Consensus estimates from S&P Global
The EPS miss was entirely driven by the ROCTAVIAN inventory write-off of $119 million (~$0.46 per share after tax). Excluding this charge, Non-GAAP EPS would have been approximately $0.92, significantly above consensus.
Full Year 2025 delivered strong growth:
What Changed From Last Quarter?
Three key developments since Q3 2025:
-
ROCTAVIAN Withdrawn — After announcing divestiture efforts in October 2025, BioMarin was unable to find a buyer and made the strategic decision to voluntarily withdraw ROCTAVIAN from the market. This resulted in ~$240 million in total charges including inventory write-off and asset impairments.
-
Amicus Acquisition Financing Complete — BioMarin secured $3.7 billion in non-convertible debt at favorable terms to fund the $5.2 billion Amicus acquisition, expected to close Q2 2026.
-
BMN 333 Advancing — Phase 1 PK data for the long-acting CNP exceeded targeted free CNP exposure levels, supporting potential to become new standard of care in achondroplasia. Registration-enabling Phase 2/3 study planned for 1H 2026.
How Did Segments Perform?
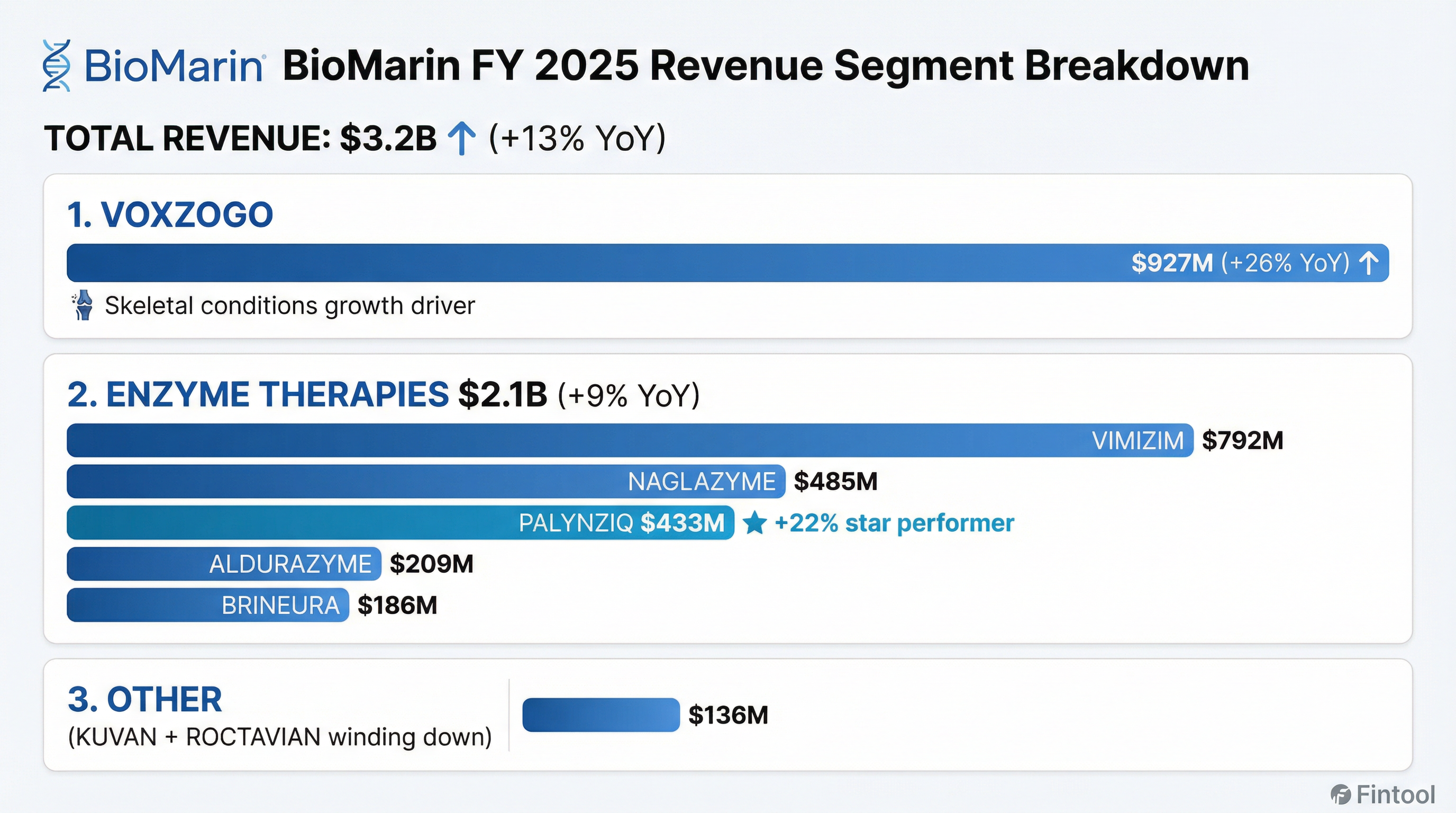
VOXZOGO: Growth Engine Accelerating
VOXZOGO delivered the strongest quarterly growth rate of the year:
- 5,000+ children now being treated globally
- ~73% of revenue from OUS markets (~$680M of $927M FY 2025)
- ~50% of new U.S. starts in under-2-year age group in Q4
Geographic success stories highlighting global commercial capabilities:
- Asia-Pacific market: ~40% penetration within 7 months of launch
- Mid-sized European country: ~70% penetration within 12 months of launch
- Management expects under-2 age group to remain 100% Voxzogo market for "next several years"
Enzyme Therapies: Broad-Based Growth
PALYNZIQ continues to be the standout performer in Enzyme Therapies with 22% growth, driven by patients titrating to maintenance dose and strong adherence. Adolescent label expansion has PDUFA date of February 28, 2026.
Other Products: Wind-Down Mode
What Did Management Guide?
BioMarin provided 2026 guidance excluding any post-close contribution from Amicus:
Key guidance notes:
- 2026 guidance includes ~$0.25 per share of pre-close Amicus operating and interest expenses
- Non-GAAP Operating Margin expected to reach ~40% for FY 2026 (excluding Amicus)
- Other Revenue decline reflects KUVAN genericization and ROCTAVIAN withdrawal
The implied 57-63% EPS growth guidance significantly exceeds consensus expectations and reflects operating leverage as the company scales past the ROCTAVIAN overhang.
What Are the Key Pipeline Catalysts?
BioMarin outlined an ambitious 2026 pipeline calendar:
Three Major Data Readouts
Two Label Expansions Expected
BMN 333: Next-Gen Achondroplasia Treatment
Registration-enabling Phase 2/3 study to begin 1H 2026:
- 60 patients each arm: BMN 333 weekly vs. VOXZOGO active control
- 90% statistical power to detect 50% increase in annualized growth velocity vs. VOXZOGO
- Target effect size: 2.25 cm/year improvement over placebo (management calls this "conservative")
- Study design: Bayesian approach allows protocol modification based on Phase 2 interim data
- Safety profile: Preclinical models show no additional safety concerns vs. Voxzogo; higher CNP exposures possible with 333 formulation
- Goal: Establish BMN 333 as new standard of care with potential "best-in-disease" efficacy profile
How Did the Stock React?
The stock's negative reaction despite the revenue beat likely reflects:
- ROCTAVIAN finality — The gene therapy chapter is now definitively closed with full market withdrawal
- EPS optics — The -18% EPS miss headlines poorly despite being non-recurring
- Amicus uncertainty — Guidance excludes Amicus contribution, leaving the combined company outlook unclear
What About the Amicus Acquisition?
The $5.2 billion acquisition announced December 2025 remains on track:
Products Being Acquired:
- Galafold (Fabry disease) — Estimated fair value ~$3.0 billion
- Pombiliti + Opfolda (Pompe disease) — Estimated fair value ~$1.6 billion
The deal is expected to significantly accelerate revenue growth and increase profitability once closed.
Historical Beat/Miss Record
BioMarin has delivered 7 consecutive revenue beats before Q3 2025:
*Consensus estimates from S&P Global
Note: Q3 2025 EPS included $221M Inozyme acquisition charge; Q4 2025 EPS included $119M ROCTAVIAN inventory write-off.
Q&A Highlights: Competitive Positioning and BMN 333
On FGFR3 Inhibitor Competitive Data
When asked about recently released infigratinib data, Chief R&D Officer Greg Friberg noted the competitor's results were "generally comparable to other CNP class effects at one year" but emphasized Voxzogo's differentiated evidence package:
"Voxzogo has a tremendous amount of supporting evidence behind it. We talk about the 10,000 patient years of safety. We also have a deep wealth of information with regard to evidence beyond height, whether it's for Foramen Magnum, physical function, tibial bowing, quality of life."
On Patient Switching Dynamics
Chief Commercial Officer Kristen Hubbard addressed concerns about patients switching to competitors:
"What we hear disproportionately is that above all else, efficacy and safety are the highest priority, convenience is the third. The fact that we have both long-term safety and efficacy data, but also importantly, durability... We are showing data now that shows that Voxzogo continues to work year-over-year, data that we published going 7 years out."
Market research indicates most physicians and patients are unlikely to switch from Voxzogo in the near term, especially given limited long-term data for competitors.
On BMN 333 Phase 3 Design
The Phase 3 portion will enroll 60 patients per arm with 90% statistical power to detect a 2.25 cm/year growth velocity improvement versus Voxzogo — which management characterized as a "floor, not a ceiling":
"We wanted to share with you what our ambition was, what we thought was not only statistically significant, but clinically meaningful... Animal model data, as well as our dose-response model, suggests that any one of the doses we're putting into phase two could potentially deliver the profile we're looking for."
The study uses a Bayesian approach, allowing protocol modifications based on Phase 2 interim data.
On Hypochondroplasia Opportunity
Management provided color on the hypochondroplasia market opportunity:
- Total addressable patient (TAP) pool: ~14,000 patients globally
- Prevalence: Similar to achondroplasia, but diagnosis rates are much lower
- Pre-launch activities: Educating on signs/symptoms, establishing referral guidelines
- Expected approval timing: 2027 (data 1H 2026, submission 2H 2026)
"The enrollment rate of the trial in and of itself shows the excitement level that could be here for hypochondroplasia."
On 2026 Quarterly Dynamics
CFO Brian Mueller warned investors about Q1 2026 expectations:
- Q1 2026 expected to be lowest revenue and EPS quarter of the year
- Both total revenue and Voxzogo revenue expected "on par with Q1 2025"
- Most pre-close Amicus costs (~$0.25/share) will hit Q1
- Revenue expected to be backloaded to Q4, similar to 2025 pattern
On Market Access Renegotiations
CEO Alexander Hardy addressed routine reimbursement negotiations in two markets:
"When you get into five years on the market, and the product is very well established... you tend to go under formal reimbursement processes. This represents actually an opportunity for us to broaden the access in those countries... you offset potentially a reduction in price, but with the opportunity to significantly expand the population."
Key Takeaways
-
Revenue momentum intact — Q4 beat by 4.8% and FY 2025 grew 13% YoY to $3.2 billion
-
VOXZOGO accelerating — 31% Q4 growth with 5,000+ patients globally; path to $1B+ in 2026
-
ROCTAVIAN chapter closed — Voluntary market withdrawal and ~$240M in charges mark the end
-
2026 EPS guidance impressive — $4.95-$5.15 implies 57-63% growth, even excluding Amicus
-
Rich pipeline calendar — 3 major data readouts, 2 label expansions, BMN 333 Phase 2/3 start in 2026
-
Amicus transformational — $5.2B deal adds Galafold and Pombiliti + Opfolda; close expected Q2 2026
-
Competitive moat intact — Management confident in Voxzogo's 10,000+ patient-years of safety data and under-2 age group exclusivity for "next several years"
-
BMN 333 ambitious targets — Phase 3 powered to show 2.25 cm/year growth improvement vs Voxzogo, with potential upside
Full earnings call transcript available at BMRN Transcripts